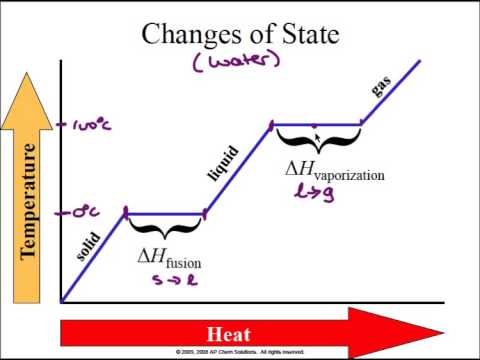
As time progresses, the magnitude of the forward rate diminishes and that of the reverse rate increases. At the start of the reaction, there is only a forward reaction since there are no products yet to react. The Upper graph is a comparison of the magnitudes of the rates of forward and reverse reactions in the system as it approaches equilibrium. 
1: shows measurable physical parameters for a system as it approaches equilibrium, starting from a conditions of only reactants and no products. Thus, although the reaction continues unabated, we see no overall changes to the amount of either A or B.įig. At this point and until some stress is imposed on the system, reactants A are being produced and used up at equal rates and so are products B. eventually, a point will be reached where the forward and reverse rates are equal. Similarly, as the amount of B is increased due to the forward reaction, then the backward rate will increase from zero. Therefore, as the amount of A is diminished from the initial situation (used up in the forward reaction) the forward rate will drop. The Kinetic Molecular theory tells us that the rate of reaction depends somewhat on the collision frequency and that the collision frequency of molecules A with each other depends on how many molecules there are in the container. If there is initially, no B then there can be no reverse reaction. The equilibrium between reactant A and product B: A ⇄ B Suffice it to say, that most times, if the concentration of reactants goes up, the rate of conversion of these reactants to products goes up too. The relationship between the amount of material and the rate of reaction is given in the section on Kinetics, later in these notes. Generally, the more the amount of reactant, or product, the faster will be the speed with which the forward, or reverse, reaction occurs. It’s not the amount that is the deciding factor. This means that some equilibrium systems can have mostly reactants, others, mostly products and in rare cases, the same amount of reactants and products. Note that there is no reference to the amounts of reactants necessary to achieve this state. Higher pressures (by compressing the container containing the reaction mixture) will favour the production of product (colourless) at the expense of the amount of reactant (coloured) hence, the equilibrium can be observed to change as the reaction vessel is compressed or expanded (as in a piston moving up and down in a glass cylinder.) Definition: Equilibrium: As it applies to a chemical reaction system: State of a reaction mixture at which the forward reaction rate is equal to the reverse reaction rate. Both temperature and pressure affect this equilibrium. Reactants are reddish brown, products are colourless. We will start with gas phase equilibria as a simple reaction type but the general principles we explore here will still hold in other phases.Ĭonsider the gas phase chemical system represent by the following chemical equation: Equilibria can occur in solid, liquid or gas. We will be exploring the more general cases where equilibrium is occurring. If the container is open and the gas is allowed to blow away then there can never be a reverse reaction as that particular component will be gone. For example, a liquid reaction mixture where one of the products is a gas. In an open system, some reactions can go to completion if it is arranged such that the product leaves the system as it is produced.Sometimes that equilibrium state may be such that the container has almost no products or almost no reactants but it is still an equilibrium. Any chemical system in a closed container will always reach a state of equilibrium. 
There is no ‘pure’ substance, no ‘complete’ reaction, in a closed container but we can often make such an assumption with little or no additional uncertainty. This assumption is not always valid and in reality, is never 100% true. 
Often (in our Stoichiometric calculations), we consider that reactions go to completion, i.e., that one or more of the reactants is completely used up.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
